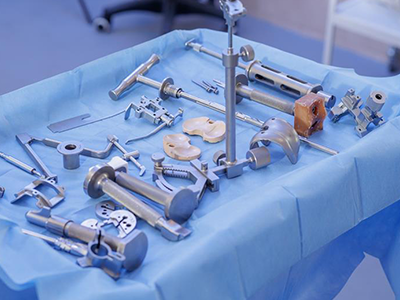
Medical Ultrasonic Cleaning Equipment
Ultrasonic Cleaning Equipment for Medical Precision Applications
Engineered ultrasonic cleaning systems for medical device manufacturing environments, including instruments, implant components, and device assemblies requiring controlled, high-precision contaminant removal during production. Designed to maintain surface integrity while ensuring consistent cleaning outcomes in regulated manufacturing and cleanroom environments.
Medical manufacturing environments require extremely controlled cleaning processes where even trace contamination can impact device safety, performance, and downstream sterilization outcomes. Conventional cleaning methods often struggle to reach micro-features, lumens, and internal geometries commonly found in medical instruments and devices during production. Incomplete cleaning can affect sterilization effectiveness, compromise device reliability, and increase compliance risks in regulated manufacturing environments.
Ultrasonic cleaners for medical instruments enable precise removal of contaminants from complex geometries without damaging delicate surfaces. The result is consistent, repeatable cleaning performance that supports safe medical use and reliable manufacturing outcomes. Zenith Ultrasonics delivers medical ultrasonic cleaning systems designed to support medical device manufacturers and precision engineering facilities with process-driven cleaning solutions built for critical production applications, not end-user or hospital cleaning processes.
Medical Cleaning Challenges
Cleaning requirements in medical device manufacturing facilities are defined by precision, repeatability, and process control, rather than scale alone. Components must meet stringent cleanliness standards before final assembly, packaging, or sterilization.
- Complex internal geometries: Internal cavities, lumens and intricate features can trap machining residues, polishing compounds and particulates, making them difficult to clean using conventional methods.
- Inconsistent manual processes: Variability in manual cleaning can lead to inconsistent results, which are unacceptable in controlled and validated manufacturing environments.
- High-volume batch processing: Large production batches require uniform and repeatable cleaning performance without compromising throughput or process efficiency.
- Regulatory process compliance: Manufacturing workflows must comply with strict regulatory requirements, where process validation, documentation, and consistency are critical.
- Repeatability and quality assurance: Consistent cleaning results across cycles are essential to ensure product quality, reliability, and compliance with medical device manufacturing standards.
How Zenith Ultrasonics Solves These Challenges
Zenith Ultrasonics designs systems that use controlled ultrasonic energy to address contamination challenges in medical device manufacturing environments without relying on mechanical abrasion or manual intervention.
- Deep-access ultrasonic cavitation: Ultrasonic cavitation in a medical ultrasonic cleaning system generates localized cleaning action that penetrates internal geometries, removing contaminants from areas unreachable by traditional methods.
- Non-contact surface protection: Sensitive medical surfaces remain unaffected due to the non-contact nature of the cleaning process, ensuring integrity of instruments and implants.
- Controlled process stability: Process stability is achieved through tightly controlled operating parameters, ensuring predictable and repeatable cleaning outcomes.
- Multi-stage cleaning workflows: Structured cleaning workflows can be implemented using sequential processing stages based on application requirements in a medical device manufacturing line.
- Reduced operator dependency: Systems are designed to minimize operator dependency while maintaining consistent cleaning quality across repeated cycles and production loads.
Common Medical Contaminants Addressed
Medical components accumulate a variety of organic and manufacturing-related residues depending on their production and usage environment.
- Manufacturing polishing compounds: Residual polishing compounds and finishing media from implant and medical device manufacturing processes remain embedded on surfaces if not properly cleaned.
- Machining oils and lubricants: Lubricants, cutting fluids, and machining oils used during fabrication of precision medical components create persistent surface contamination.
- Microscopic particulate contamination: Fine particles become trapped within tight tolerances, micro-features, and internal geometries of medical components.
- Chemical surface films: Residual chemical films from surface treatment, passivation, or cleaning processes can affect device performance and sterilization readiness.
Typical Medical Components Cleaned
Zenith Ultrasonics systems are designed for medical device manufacturing environments, supporting precision cleaning at various stages of production. These systems are used to remove machining residues, oils, particulates, and other contaminants to meet stringent industry standards before final assembly, packaging, or sterilization.
Applicable Zenith Ultrasonic Systems
Zenith Ultrasonics offers a comprehensive range of ultrasonic cleaning systems engineered for medical device manufacturing environments. These systems are designed to remove machining oils, particulates, and process residues from components and assemblies, supporting critical cleaning requirements prior to final assembly, packaging, or sterilization.
- Benchtop Ultrasonic Cleaners: Benchtop ultrasonic cleaners from the MEC2 series are compact, table-top systems designed for precision cleaning of small medical components and parts during manufacturing, ensuring efficient removal of contaminants in controlled production settings.
- MEC Multi-stage Ultrasonic Cleaning Systems: MEC multi-stage ultrasonic cleaning systems are integrated multi-tank solutions that combine cleaning, rinsing, and drying processes to support validated manufacturing workflows with consistent process control and repeatability.
- STC High-Capacity Single Tank Cleaners: STC high-capacity single-tank cleaners are heavy-duty ultrasonic systems designed for larger medical components and higher production volumes, delivering strong cavitation for deep and effective removal of manufacturing residues.
- MTC Manual Multi-Stage Systems: MTC manual multi-stage systems are flexible multi-tank setups that enable sequential processes such as wash, rinse, and final rinse, making them suitable for controlled cleaning steps within medical device production lines.
- TRANSTAR II Automated Cleaning Systems: TRANSTAR II automated cleaning systems use an overhead transfer mechanism to move components through multiple cleaning stages, making them ideal for cleanroom manufacturing environments requiring repeatable and contamination-controlled processing.
- Ultramatic One-Tank Automated Systems: Ultramatic one-tank automated systems perform cleaning, rinsing, and drying cycles within a single integrated unit, offering efficient and streamlined processing for medical components during production.
- Ultramatic HV Automated Systems: Ultramatic HV automated systems are high-performance solutions designed for demanding manufacturing applications that require precision cleaning, consistency, and high throughput.
- VORTEX Centrifugal Ultrasonic Cleaners: The VORTEX centrifugal ultrasonic cleaner combines ultrasonic energy with centrifugal motion to achieve ultra-clean, residue-free results for high-precision medical device components during manufacturing.
- ADVANTAGE Automated Ultrasonic Cleaning Systems: ADVANTAGE automated ultrasonic cleaning systems are modular multi-stage solutions designed for scalable medical device manufacturing operations, offering customizable process steps and validation-ready performance.
Application Engineering & Support
Effective medical cleaning requires understanding part complexity, contamination type, and manufacturing workflow requirements. Zenith Ultrasonics provides application-focused support to ensure optimal system performance in production environments.
- Application analysis: Evaluation of instruments and device components to identify contamination challenges and process requirements.
- Custom system configuration: Systems are designed around real workflow needs, including capacity, cycle optimization, and automation integration.
- Implementation support: Guidance during installation and early operation to ensure correct process setup and stable performance.